In brief:
- Biotech companies, enabled by private investment are a key source of new, innovative therapies and are at the heart of the life science sector
- ICG’s Life Sciences team combines seasoned drug development experience alongside company builders, to help take ideas and medicines through clinical trials to the market
- The team’s differentiated approach focuses on the needs of patients first, then finds companies with potential new drugs to deliver solutions for patients
- The profound societal impact that life science investments has, chimes with ICG’s wider ESG focus
Life sciences is a sector capable of driving profound transformation in medicine, society, and people’s lives
Major technological advances such as brain imaging and the sequencing of the human genome almost a quarter of a century ago have helped to unlock our understanding of complex diseases, their causes, and how to treat them. This includes a deeper knowledge of cancer and new insights into the workings of our brains. Research is opening the door on a transformative new generation of treatments.
Bringing life-changing treatments from theories on a chalkboard through to drugs that change lives is a complex process of clinical trials. But it’s a process that ICG’s Life Sciences team knows well.
When Dr. Allan Marchington, Head of Life Sciences at ICG, first formed his team as Bridge Valley Ventures, he sought “people who really understood how to develop drugs and had experience of doing so”. But also, he adds, “people who’ve been CEOs, who’ve built businesses and worked with investors”.
With their collective experience, the Life Sciences team can focus on patients’ needs and develop treatments that make a truly substantial impact to people’s lives and wellbeing. With the world’s population forecast to reach 9.7 billion by 2050, 17% of whom will be over 65, there’s a growing need to unlock new treatments that can change lives and society.

Combining experience in science and in building companies
The life science sector’s grounding in academia means experimentation and learning from results are important factors in making investments.
“We’ve all got an investment track record, and we all know how to develop drugs to get them through to the clinic and into the market,” says Simon Tate, Managing Director of Life Sciences at ICG. “We can expertly and efficiently do due diligence to assess assets because of that experience as well. That’s one of our real USPs.”
That experience is also something ICG looks for in a potential investment. Just as investors look for founders who have launched and sold businesses before, teams that have successfully taken drugs through clinical trials can represent compelling investment opportunities.
We’re people who’ve been CEOs, who’ve built businesses and worked with investors.
Allan Marchington, Head of Life Sciences, ICG
A key part of successful clinical trials is collaboration, and Marchington says: “We look for a management team that works well together and is upfront and transparent about what’s being done.“
“Because a lot of these firms are small, you need to be able to have these honest conversations,” he adds. “Typically, our investments have a three-to-six-year lifespan, so we’re building a long-term relationship and it’s a relationship in which we need to add value not just capital. The team’s collective hands-on healthcare experience allows us to add tremendous value to our portfolio companies.”
Ultimately, however, the patient remains the focus of the Life Science’s team philosophy – delivering real change that makes a difference in patients and their carers’ lives, in an accessible and affordable way.
“A treatment has to really move the needle. I think investors and pharma companies know it’s no longer acceptable to just have incremental improvements,” says Tracy Weightman, Associate Director of Life Sciences at ICG. “It has to address something that’s currently missing in patient needs or care, and really impact either the quality of life or their life expectancy.”
ICG Life Science’s investment in Wugen Therapeutics, is an example of this thinking in action. The company began life in Washington University in St. Louis and uses the emerging science of cell therapy to harness the human body’s memory natural killer (NK) cells and enhance their tumour-killing functionality.
The investment enabled Wugen to advance ongoing trials for patients with severe acute myelogenous leukaemia (AML) and other oncology indications, including head and neck cancer and melanoma.
Biotech companies like Wugen are developing treatments that can help the body’s cells find and kill cancers – it sounds like science fiction to those outside the industry. But it’s just one example of how transformational companies in the sector have the potential to impact the lives of patients and wider society.
How biotechs collaborate to bring new treatments to market
The life sciences sector suddenly gained a new relevance among public consciousness at the onset of the COVID-19 pandemic. Mainstream media particularly highlighted the crucial role of collaboration between academia, biotech start-ups, and multi-national pharmaceutical companies. The Oxford/AstraZeneca and Pfizer/BioNTech collaborations are key examples of the power of this collaboration in practice.
Weightman highlights this shift in drug development and innovation approaches: “Twenty years ago, several big pharmaceutical companies did everything under their own roofs.”
“Over the past 20 years they’ve changed their business model, like a lot of other different industries and are much more collaborative. Now pharma works with biotech and academia on a regular basis to bring innovative therapies to the market.”
Within this structure private capital plays an important role in bringing visions for new treatments from the lab through to patients.
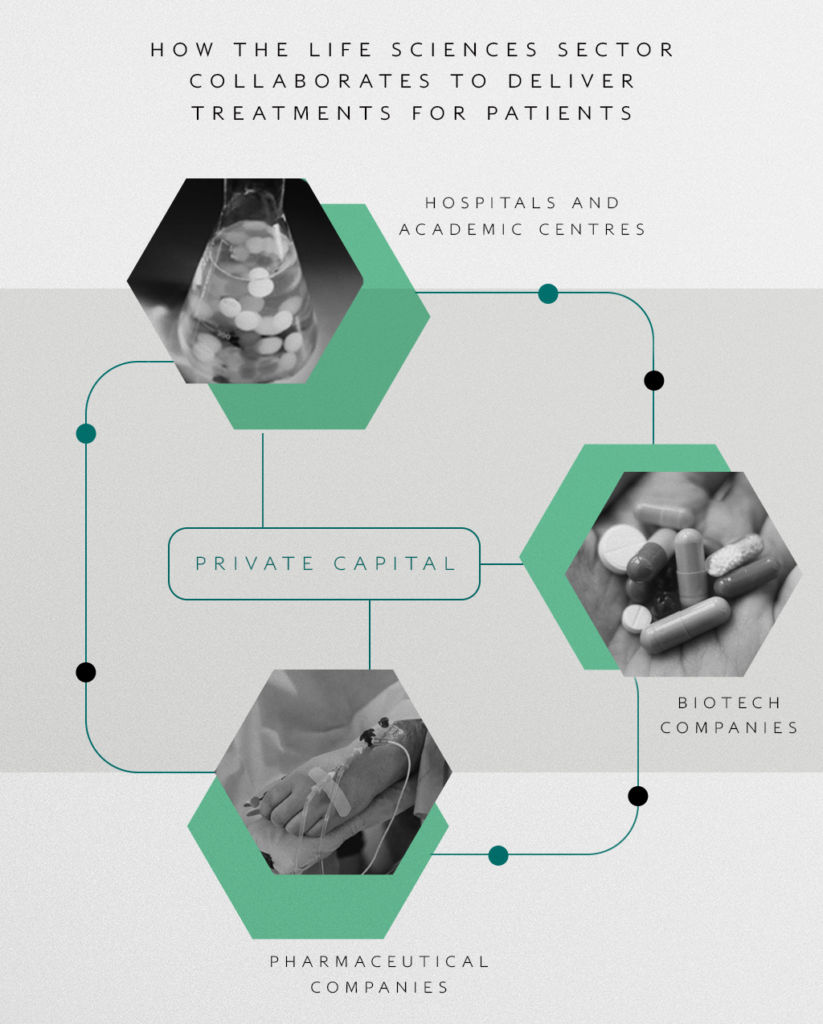
The drug development process & role of private capital
Developing opportunities within the biotech companies we support requires taking treatments through clinical trials, involving rounds of testing on progressively larger groups of patients. These trials are crucial to ascertain the effectiveness and safety of a drug to gain regulatory approval.
| Clinical trial stages | |
| Phase I 👱🏽♀️👨🏾👩🏿🦳🧔🏻 |
Tests the safety of a new treatment Looks for side effects of a treatment. Only involves a small number of people, often healthy volunteers. |
| Phase II 👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾+ |
Trials the new treatment Trials a treatment, usually, in people who have the condition for which the treatment is intended to be used. Ensures the treatment is safe and has some effect on that condition. Phase II trials also help assess what dose of treatment might be needed to be effective. |
| Phase III 👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻+ |
Trials involving larger numbers of patients Trials involving larger numbers of patients (hundreds or sometimes thousands), who are usually randomised to receive the new treatment, the best available current treatment, or a placebo. The aim is to assess how well the new treatment works. |
| Phase IV 👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾👩🏿🦳🧔🏻👱🏽♀️👨🏾+ |
Trials are done after a drug has been approved Post-approval trials are carried out to gather information on the drug’s effect in various populations, and any side- effects associated with long-term use. |
Sources: Medical Research Council (UK MRC); Cancer Research UK
For investors backing biotech companies in the earlier stages of development, the challenge lies in identifying ideas and teams that can bring products to market, and with whom ICG can collaborate to design trials that can win regulatory approval.
“There must be a reason to believe the hypothesis being tested has a high chance of working and will be a benefit to patients ,” says Tate. “This is where the focus on the patient becomes really important, because what we’re looking for in a company is that they understand how their new drug or therapy is going to be used by patients, and how it will change their treatment paradigm
The capability for drugs and treatments to succeed through trials and make a real difference in the lives of patients is knowns as clinical translation. It’s identifying companies capable of making it through this progression – at which point the ICG Life Science team’s experience becomes invaluable in designing capital-efficient trials and engaging with management to navigate the best route to bring treatments to patients.
Investing that improves lives and society
While in many parts of the world there is a sense that the pandemic is over, COVID-19 has a had a long-term impact on the life sciences sector and has shone a light on the importance of developing and funding new drugs.
While Oxford and AstraZeneca’s vaccine highlighted the role of collaboration across the industry, BioNTech’s RNA technology, which spurred Pfizer’s vaccine showcased a venture-backed biotech company offering a crucial solution on a global scale.
A treatment has to really move the needle. I think investors and pharma companies know it’s no longer acceptable to just have incremental improvements.
Tracy Weightman, Associate Director, Life Sciences, ICG
Biotech companies are also driving new research and innovation around neurology, a space that can offer treatments that can not only change patient and carer’s lives, but deeply affect society.
“In the ageing population there is a growing number of patients with neurodegeneration, Alzheimer’s disease, Parkinson’s disease, dementia. Right now, there are no good treatments for those patients and a lot of money has gone into that,” explains Tate.
ICG Life Science is invested in CuraSen Therapeutics, a company rethinking therapeutic approaches to treatment of these diseases. In simplified terms, the company uses drugs to turn on the pharmacology of a patient’s living cells and the nerves to supercharge them, so they work much better than before and no longer die.
We’re looking for companies that understand how their new drug or therapy is going to work in patients, not just in the lab.
Simon Tate, Managing Director, Life Sciences, ICG
“The vision here is to reset patients back to an earlier stage of their disease, perhaps three to five years,” says Tate discussing CuraSen. “They may still have aspects of the disease, but they will have a much-improved quality of life. The impact on the patient and on the carers is huge. For me, this is one of the most exciting things that I’ve done in my career, and we’re on the cusp of doing it.”
In addition to the profound impact life sciences investments can have on society, they resonate with ICG’s wider focus on ESG investing. In particular, the EU’s Sustainable Finance Disclosure Regulation (SFDR) (which sets out rules for classifying and reporting on sustainability and ESG) Article 9 type is focused on sustainability that contributes to an environmental or social objective.
ICG Life Sciences’ focus on investing in treatments for diseases where there are limited options available to patients, allow it to fit into such categorising. And in Europe where ESG is already embedded in such investing, there is ample possibility for new, transformative treatments.
Watch: Introducing the ICG Life Sciences team
Go deeper:
- Explore ICG Life Sciences
- The background to Life Sciences at ICG
- Read more about ICG Life Sciences’ investment in Wugen














 Back
Back